Home
Research Interests and Achievements
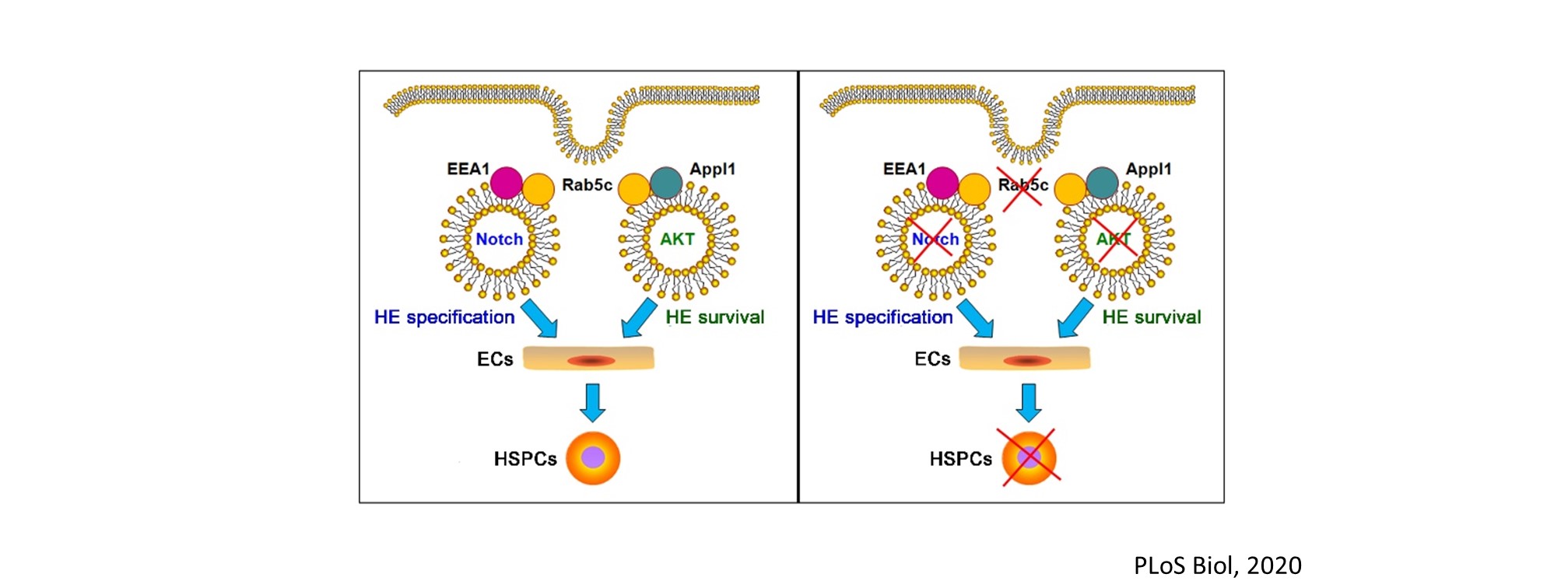
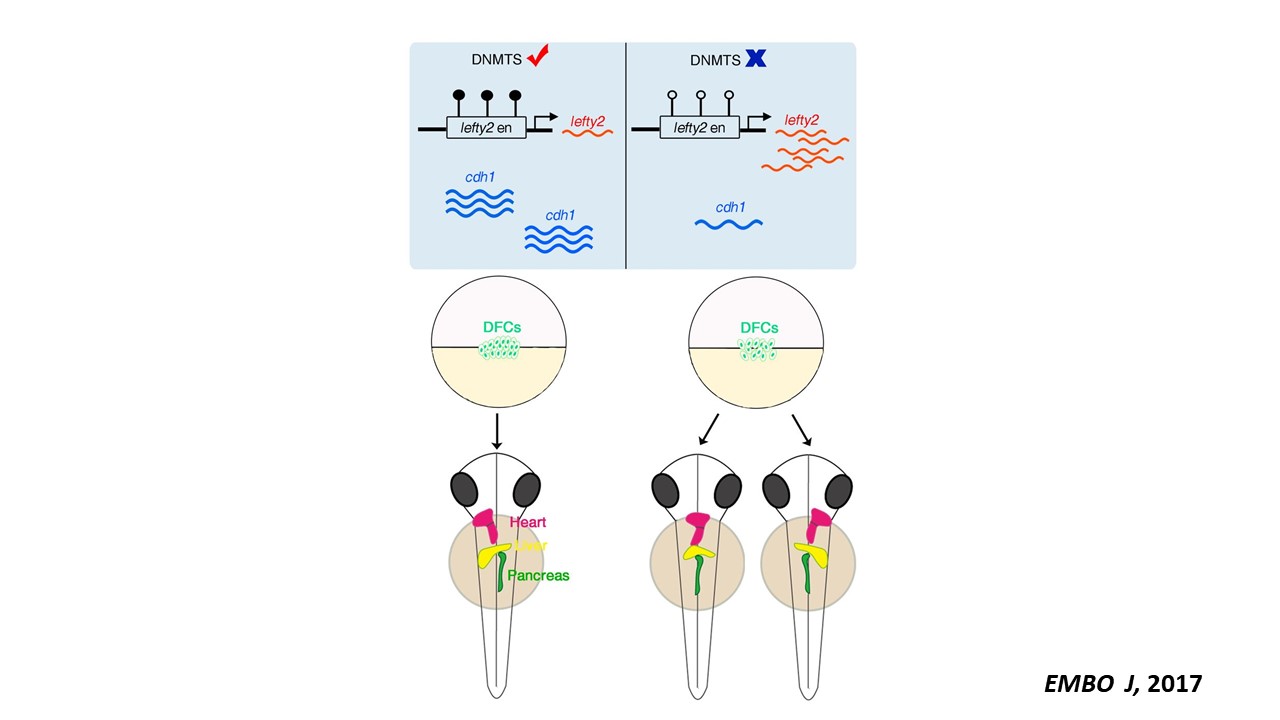
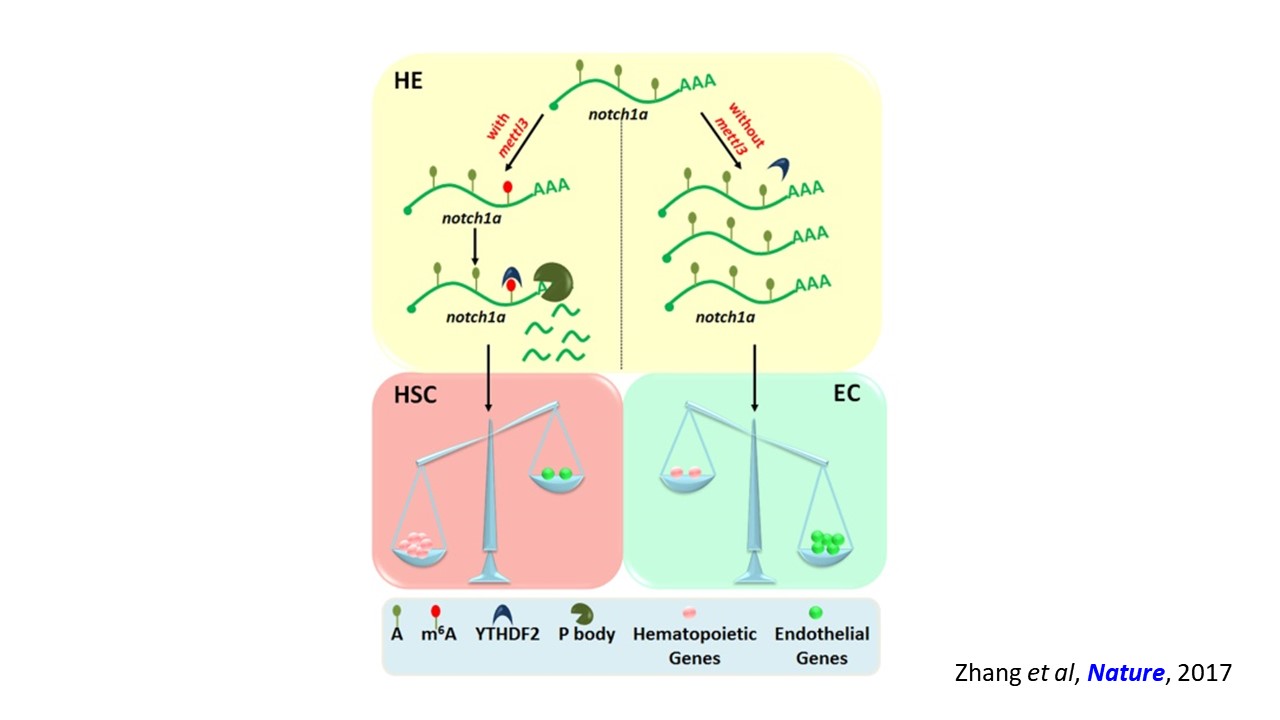
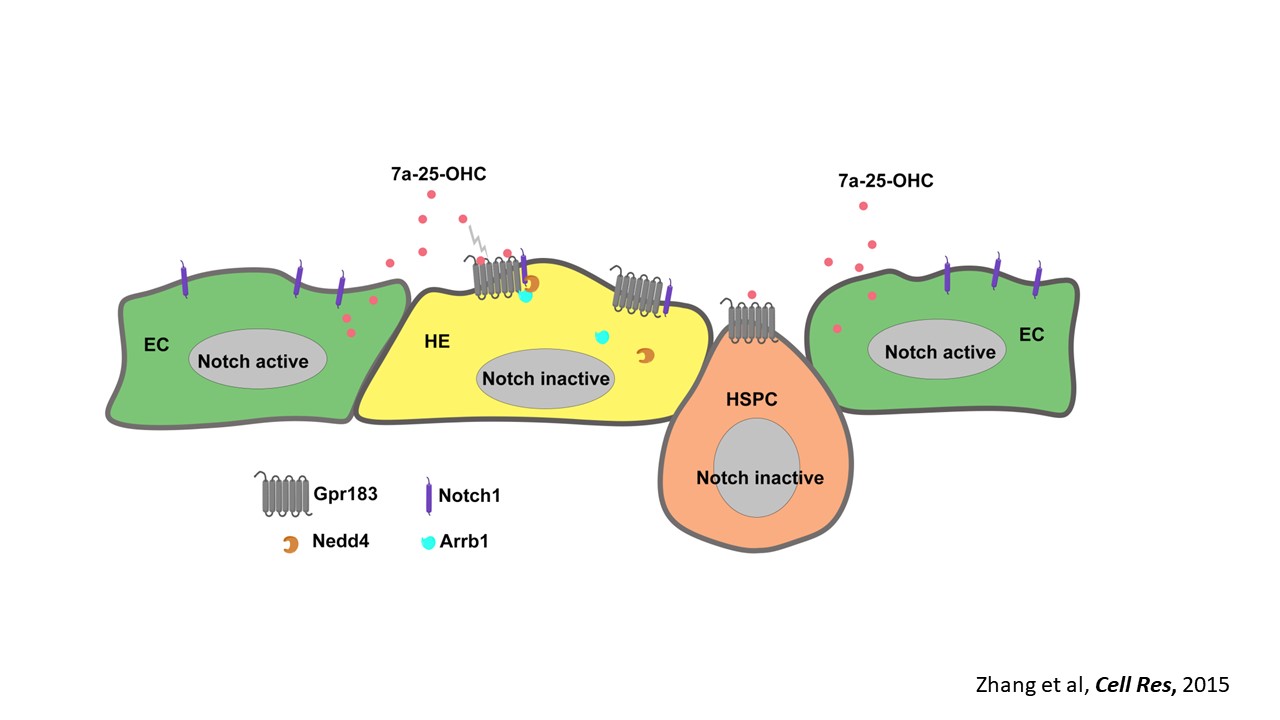
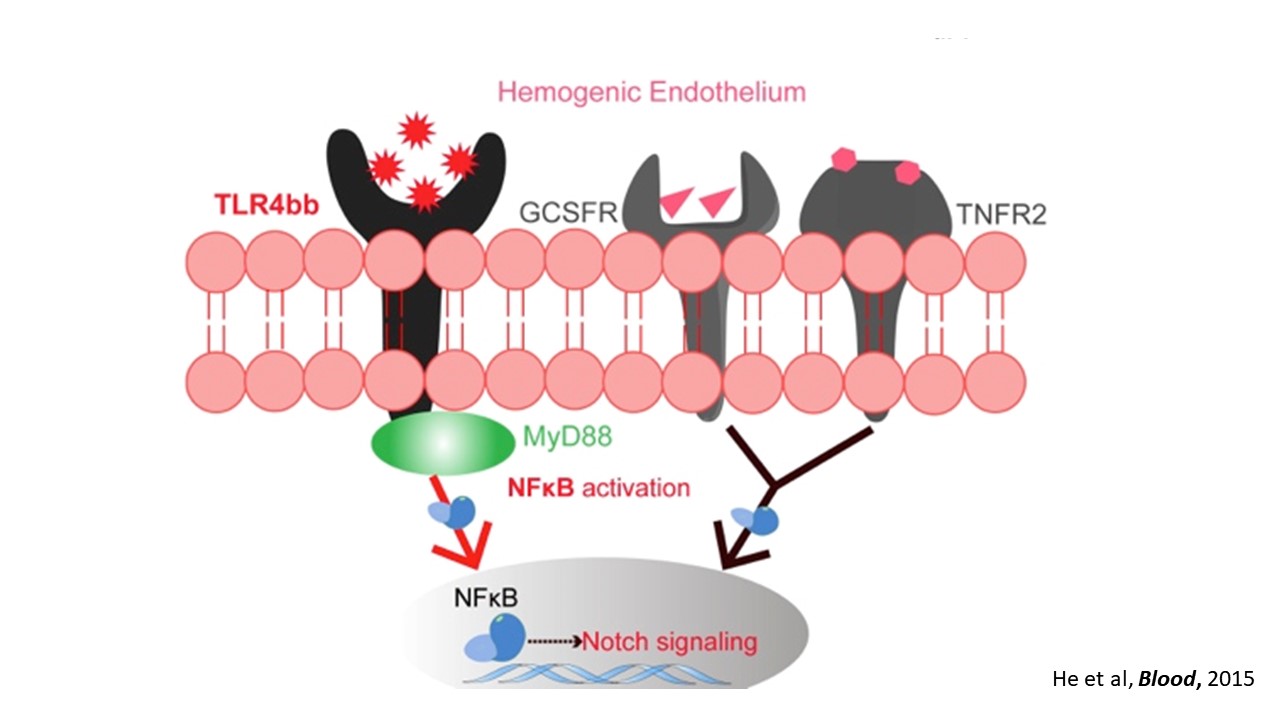
We are interested in developmental hematopoiesis in vertebrates. By employing zebrafish and mouse as model organisms, over the past decade we have focused on the transcriptional and epigenetic regulation of hematopoietic stem and progenitor cell (HSPC) development. We have demonstrated the crucial role of Notch signaling in controlling HSPC generation. Our studies highlight the dynamic requirement of Notch signaling, which is regulated by m6A modification (Nature 2017), GPR183 (Cell Res 2015), inflammatory signaling and others (Blood 2015; Blood 2014). We also revealed the indispensable roles of primary cilia (Nat Commun 2019) and endocytic trafficking (eLife 2016; PLoS Biol 2020) in transducing Notch signaling. These findings give insights into the molecular mechanisms of programming/reprogramming of HSPCs for potential therapeutic applications.
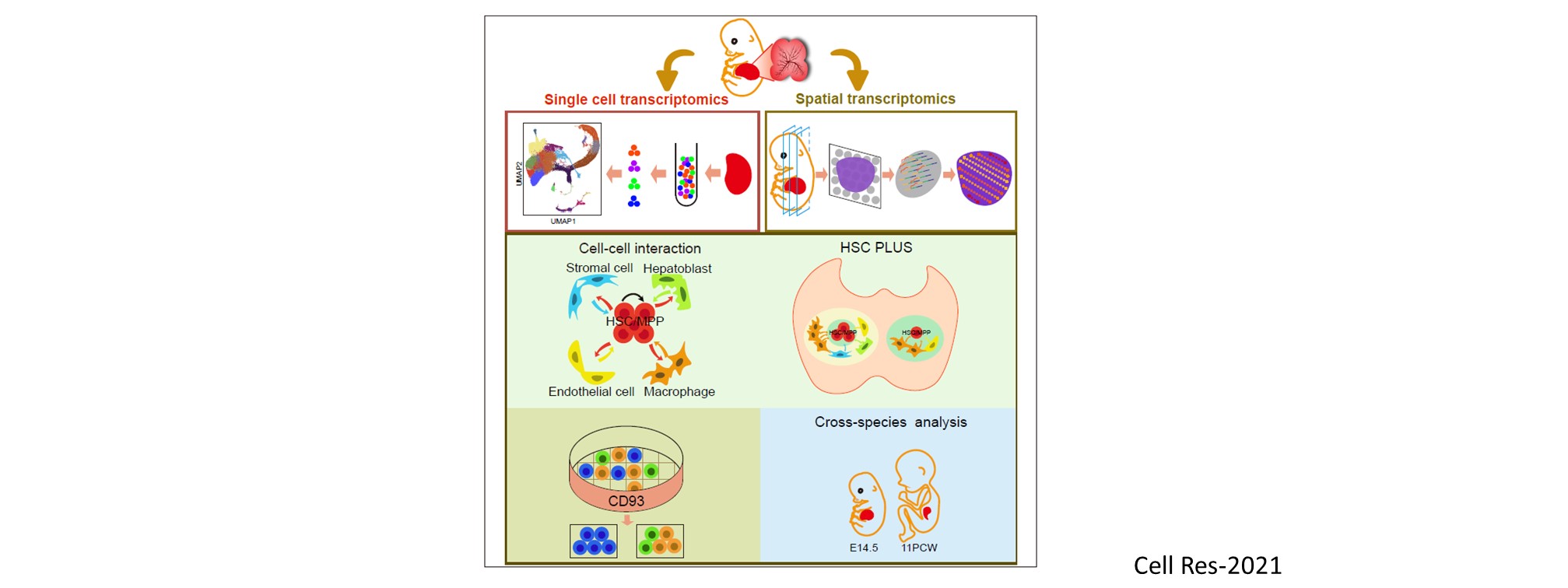
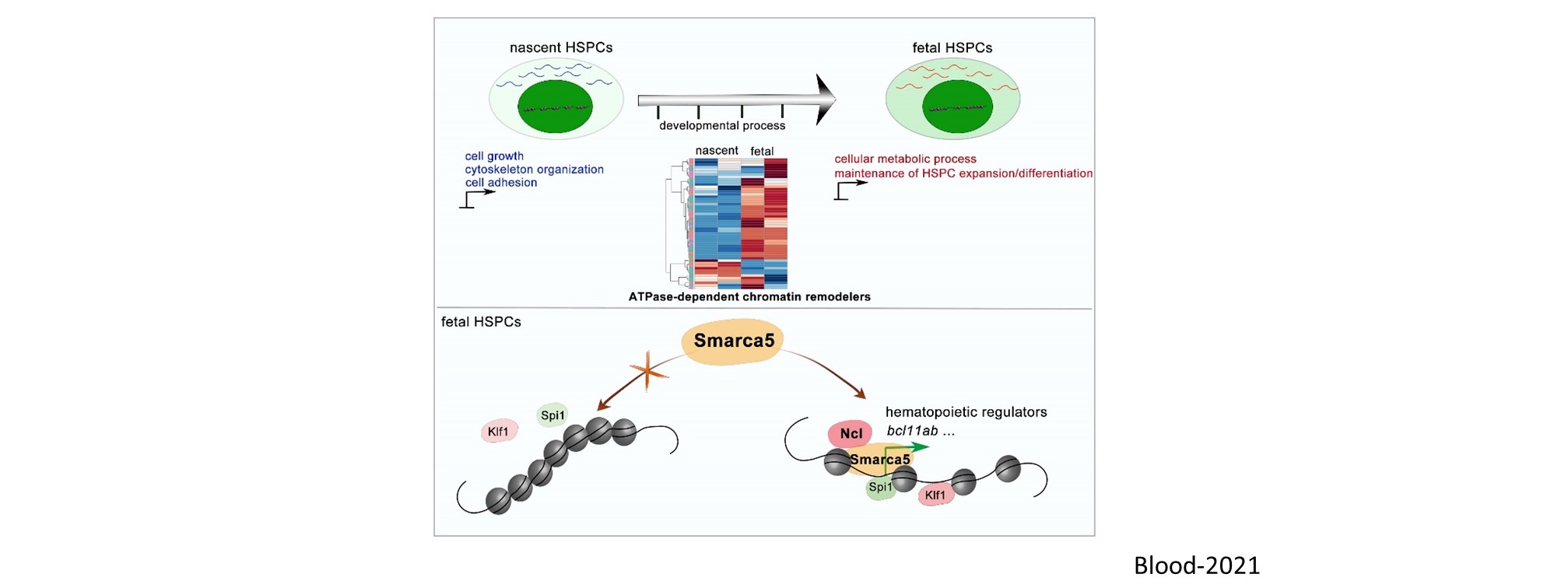
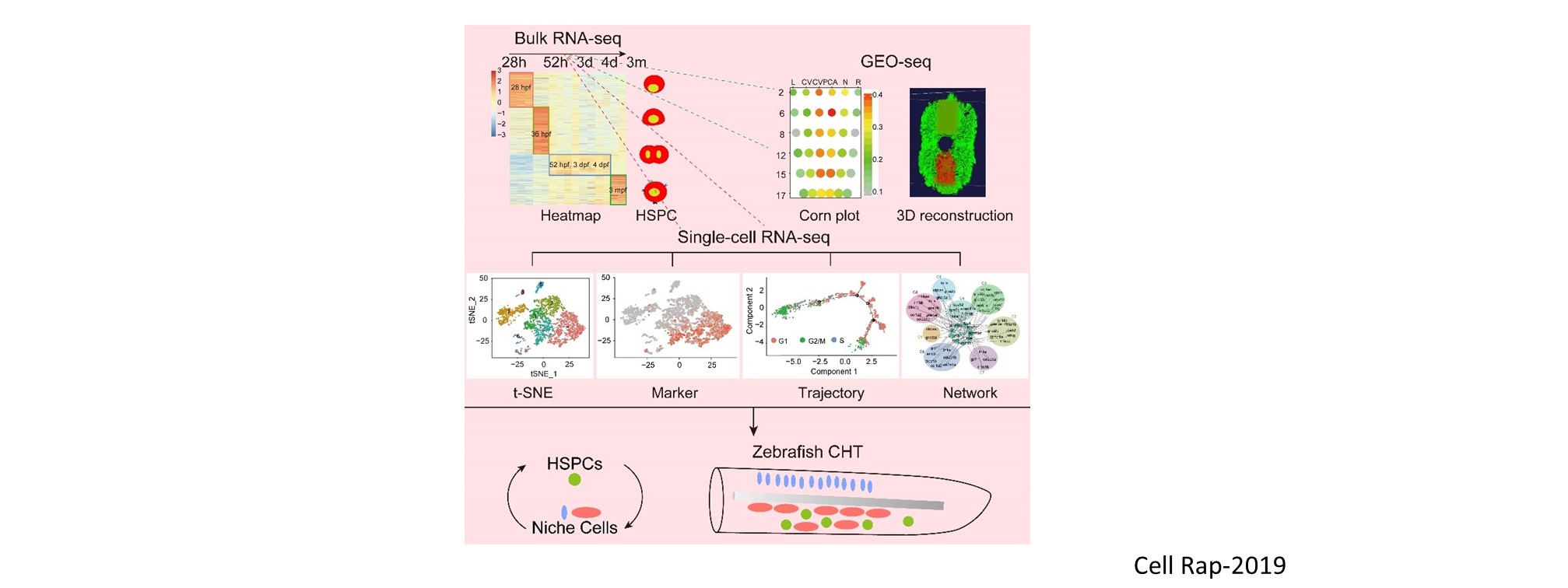
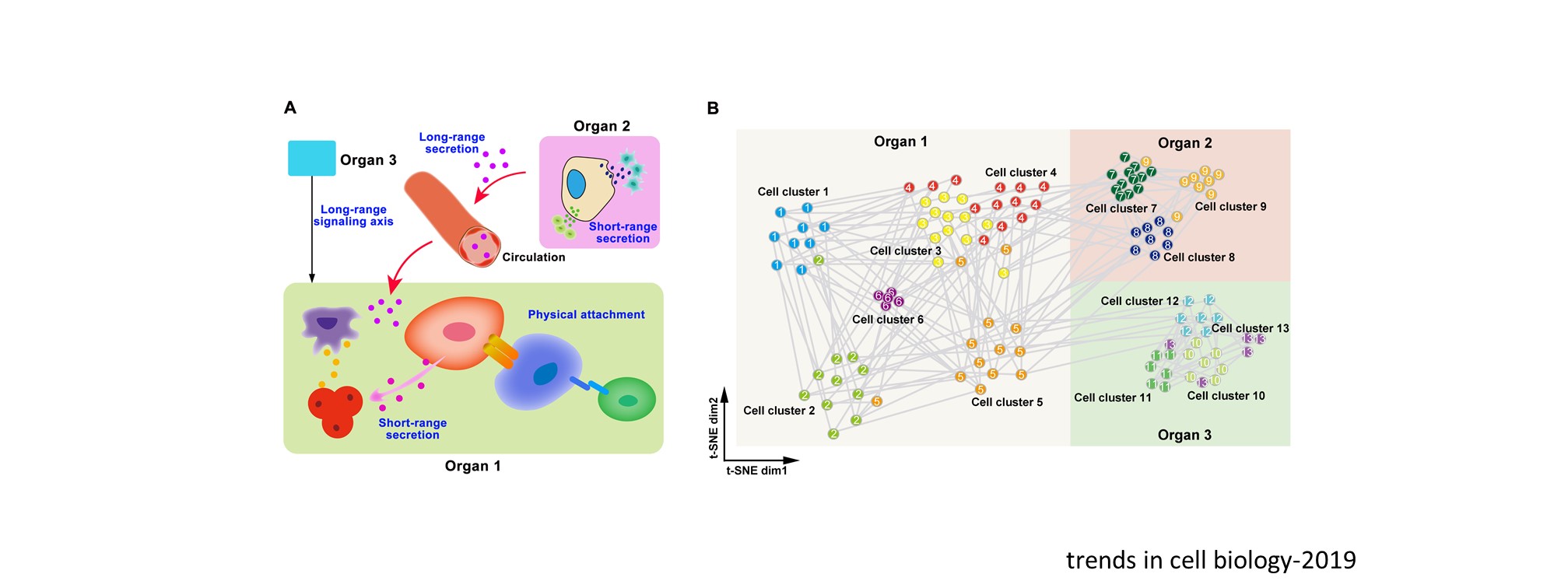
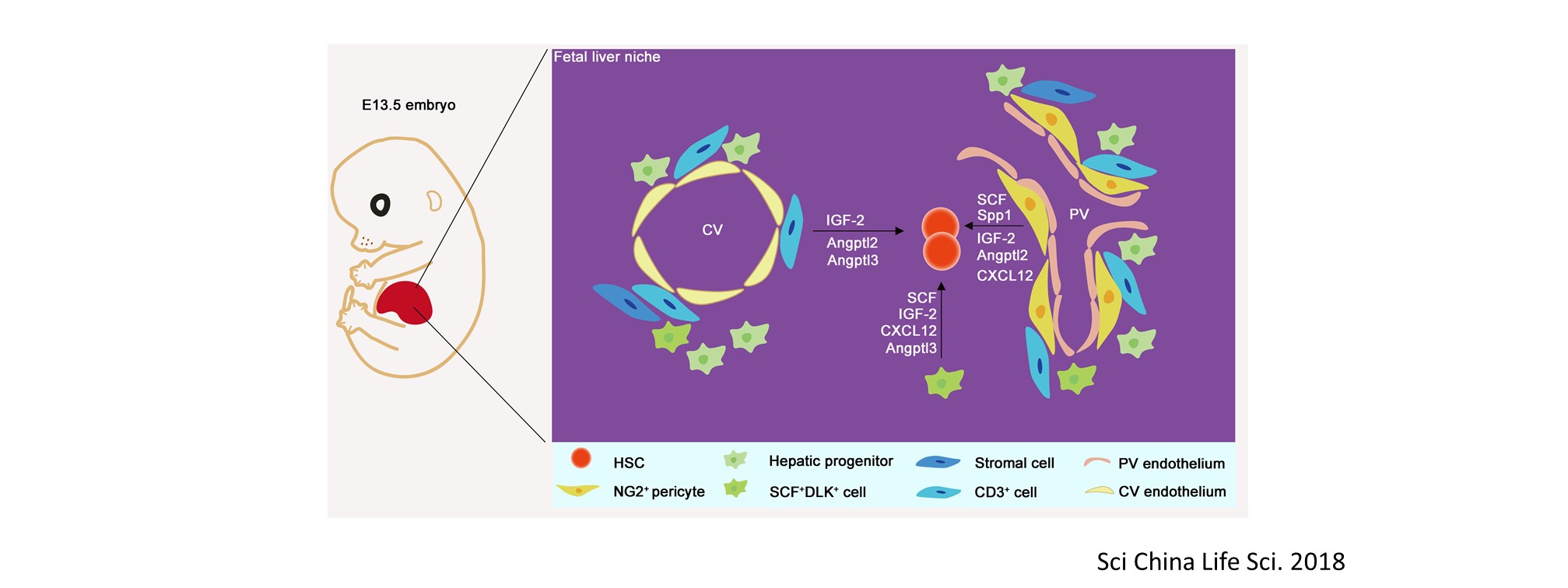
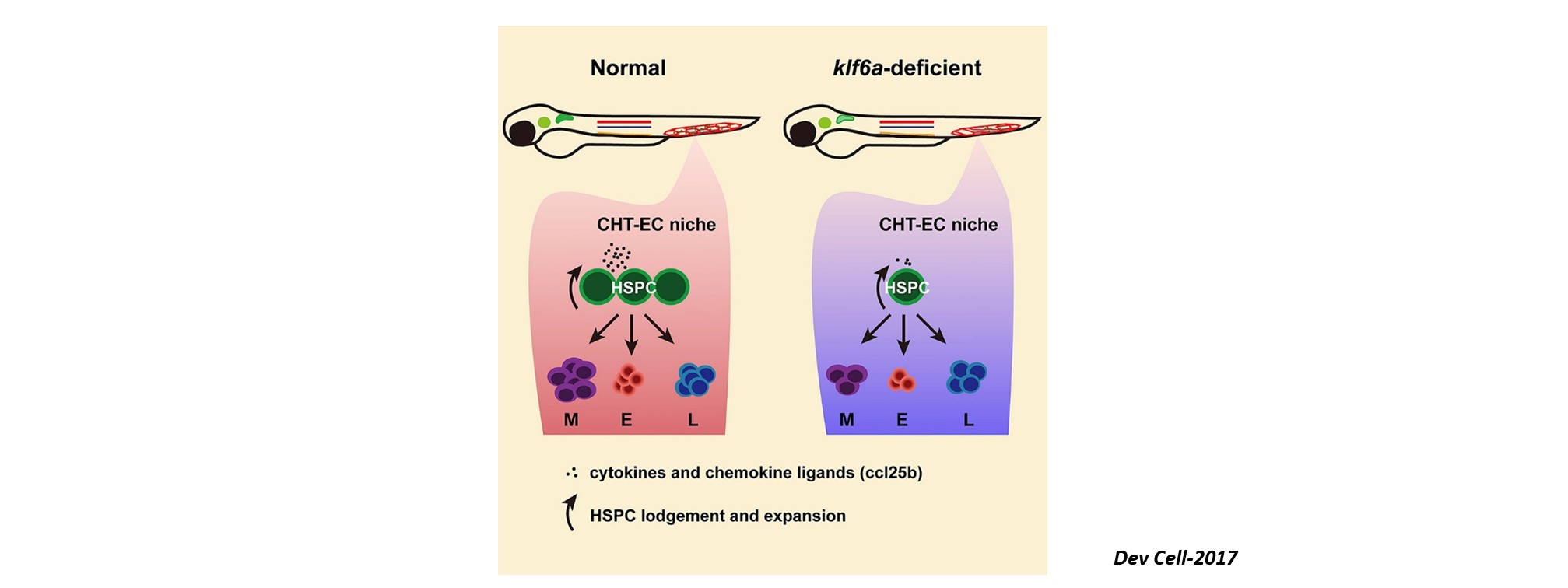
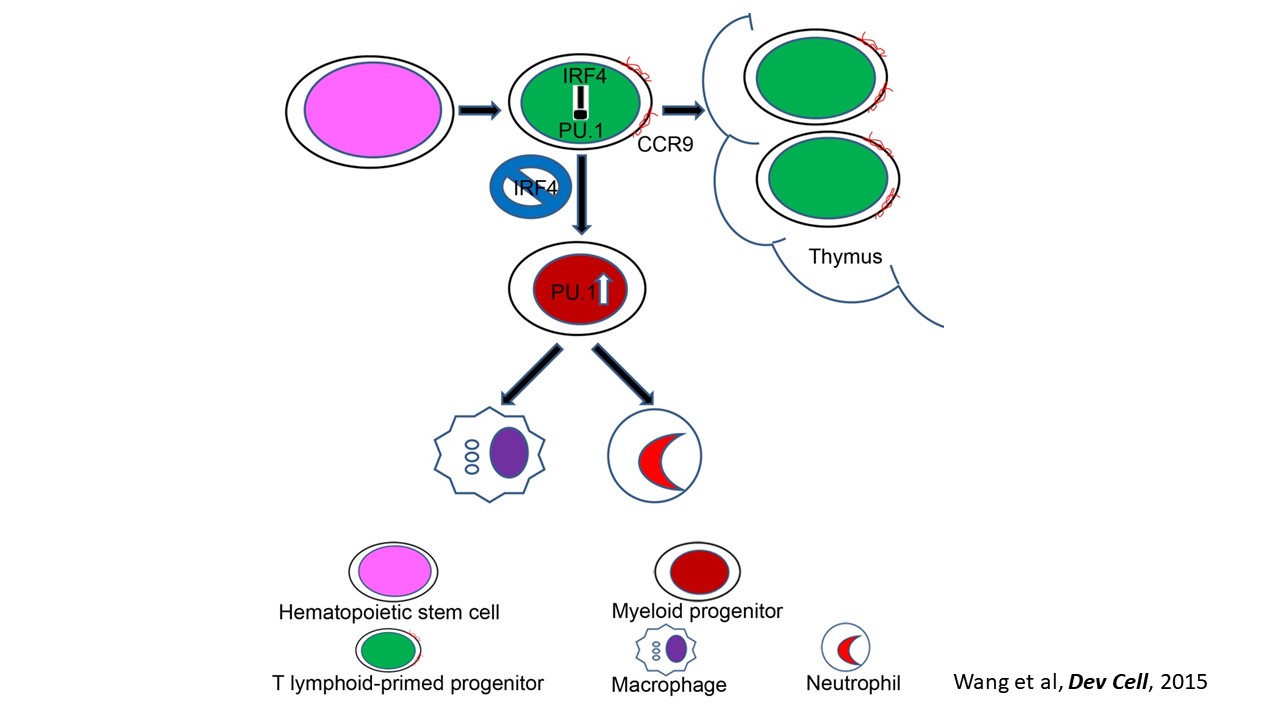
Nascent HSPCs will undergo maturation, expansion, and differentiation subsequently, and we have identified corresponding key regulators, including chromatin remodeler Smarca5 (Blood 2021), Klf6a-Ccl25b signaling (Dev Cell 2017), and transcription factor Irf4 (Dev Cell 2015, cover story). To comprehensively understand the coordinated development of HSPCs and niche, we recently use single-cell omics to profile cell atlases of developing hematopoietic organs, such as spatio-temporal transcriptome atlases of zebrafish caudal hematopoietic tissue and mouse fetal liver (Cell Rep 2019; PNAS 2021; Cell Res 2021), and temporal transcriptome atlas of mouse placenta (Dev Cell 2021). These findings will help engineer artificial niches for HSPC generation and expansion in vitro.
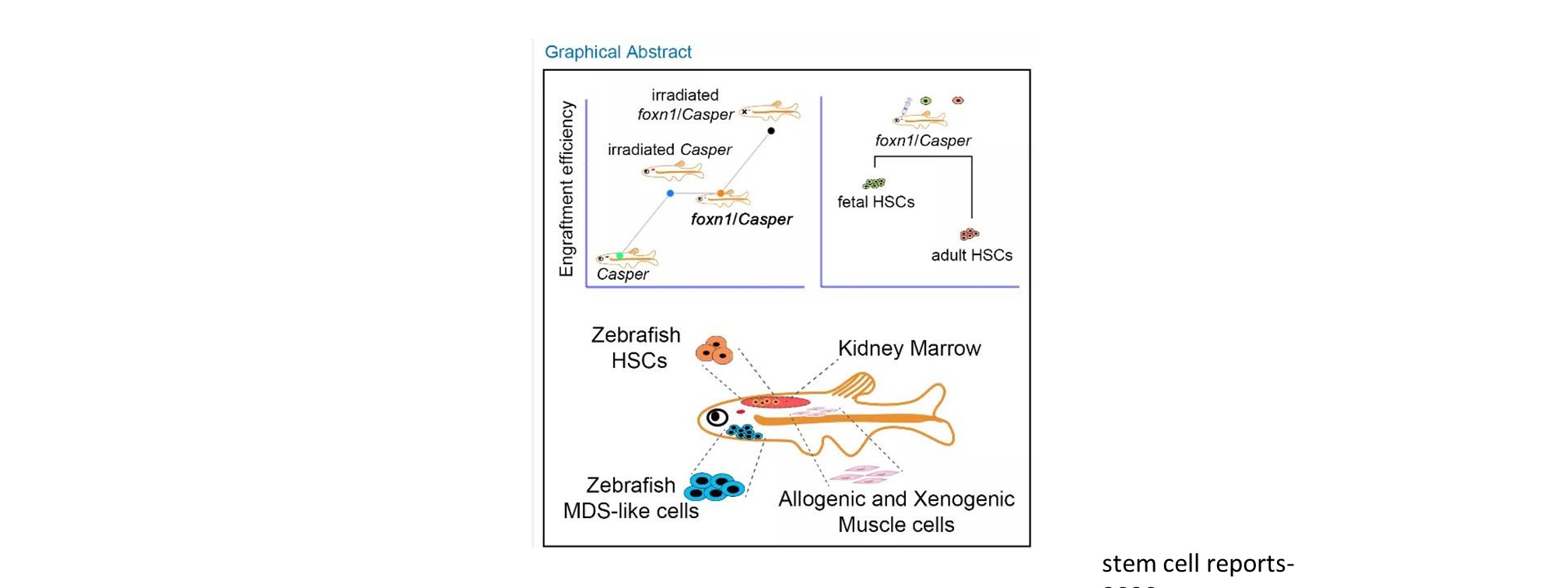
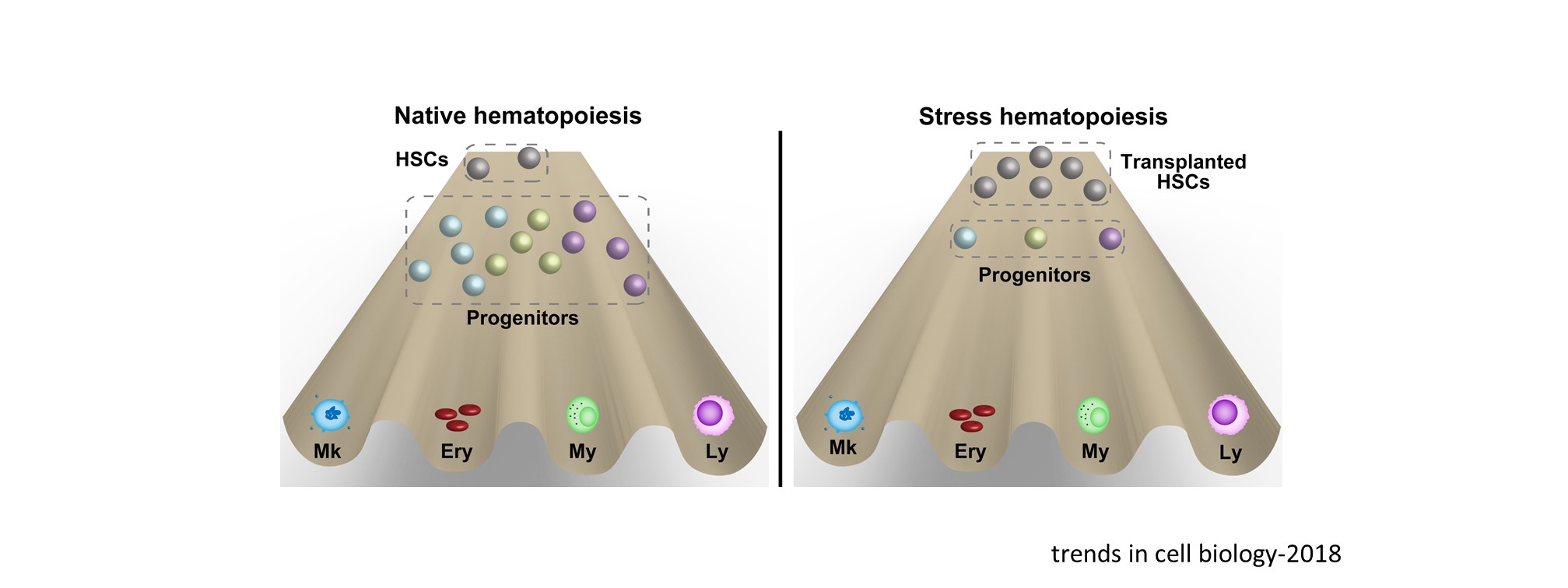
In addition to mechanistic insights, we also have generated several transplantation and disease models to visualize cellular dynamics under physiological and pathological conditions (Stem Cell Reports 2020; eLife 2021).